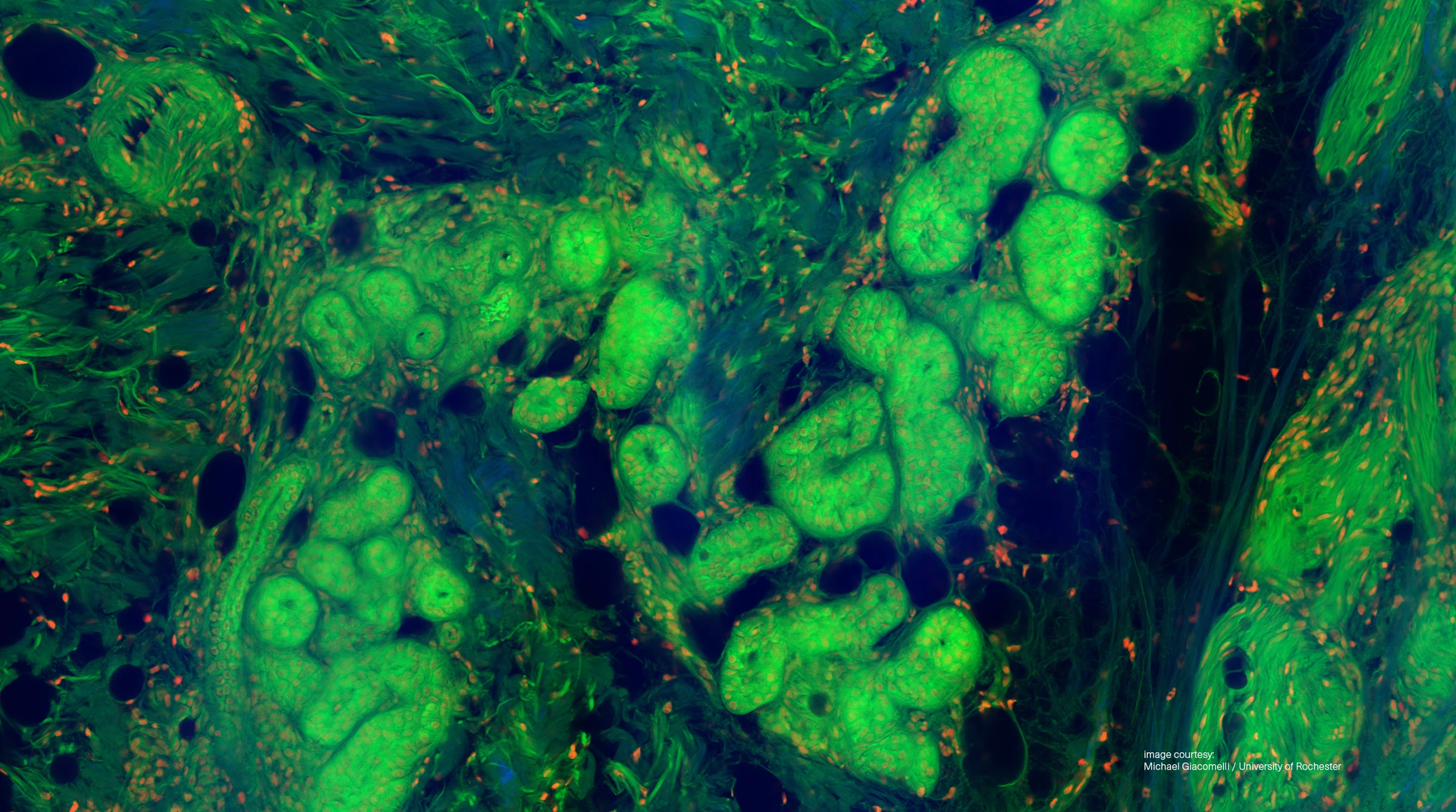
Label free & clinical imaging
In life sciences and medical research, light is key to understanding the complexity of living systems. Today, advanced laser-based technologies such as Raman and autofluorescence imaging allow scientists and clinicians to visualise molecular and structural information without the use of dyes or contrast agents. This label-free imaging technique reveals the native biochemical composition, metabolism, and morphology of tissues, cells, and biomolecules, providing real-time insights with minimal sample preparation and no phototoxicity.
Principles of label-free optical imaging
Raman spectroscopy and Raman microscopy

Raman imaging relies on the inelastic scattering of photons - the Raman effect - where a small portion of light interacts with molecular vibrations and shifts in energy. Each molecule has a unique Raman fingerprint, allowing precise chemical identification. By focusing a laser into a biological sample and detecting these shifts, Raman microscopy maps the spatial distribution of biomolecules such as proteins, lipids, and nucleic acids.
→ Typical lasers: Single frequency lasers at 405, 532, 633, or 785 nm.
Autofluorescence microscopy
Autofluorescence microscopy harnesses the intrinsic fluorescence of biological tissues to enable label-free imaging. Endogenous molecules such as NADH, flavins, collagen, and elastin emit light when excited at specific wavelengths, revealing tissue structure, metabolic activity, and biochemical composition without the need for external dyes. This minimally invasive approach preserves native biological conditions while delivering high-contrast images.
Two-photon autofluorescence microscopy
Two-photon autofluorescence microscopy extends these capabilities by using two-photon excitation with ultrafast near-infrared laser pulses. Here, fluorescence is generated only when two low-energy photons are absorbed simultaneously at the focal point. This localized excitation enables deeper tissue penetration, intrinsic three-dimensional resolution, and reduced photodamage—making it ideal for high-resolution imaging of thick samples or even in-vivo.
→ Typical lasers: Femtosecond fiber lasers at 780, 920 or 1050 nm
Second harmonic generation (SHG)
SHG imaging uses nonlinear optical interactions where two photons combine to form one photon at exactly twice the frequency (half the wavelength). This process happens naturally in structures that aren't centrosymmetric, like collagen, myosin, or microtubules. This allows us to see connective tissue and the structure of the cell's cytoskeleton clearly without using stains.
Third harmonic generation (THG) imaging is possible through the absorption of three photons.
→ Typical lasers: Femtosecond fiber lasers at 780, 920 or 1050 nm
Fluorescence lifetime imaging (FLIM)
D. Santos, Uni Ulm, GerBi workshop
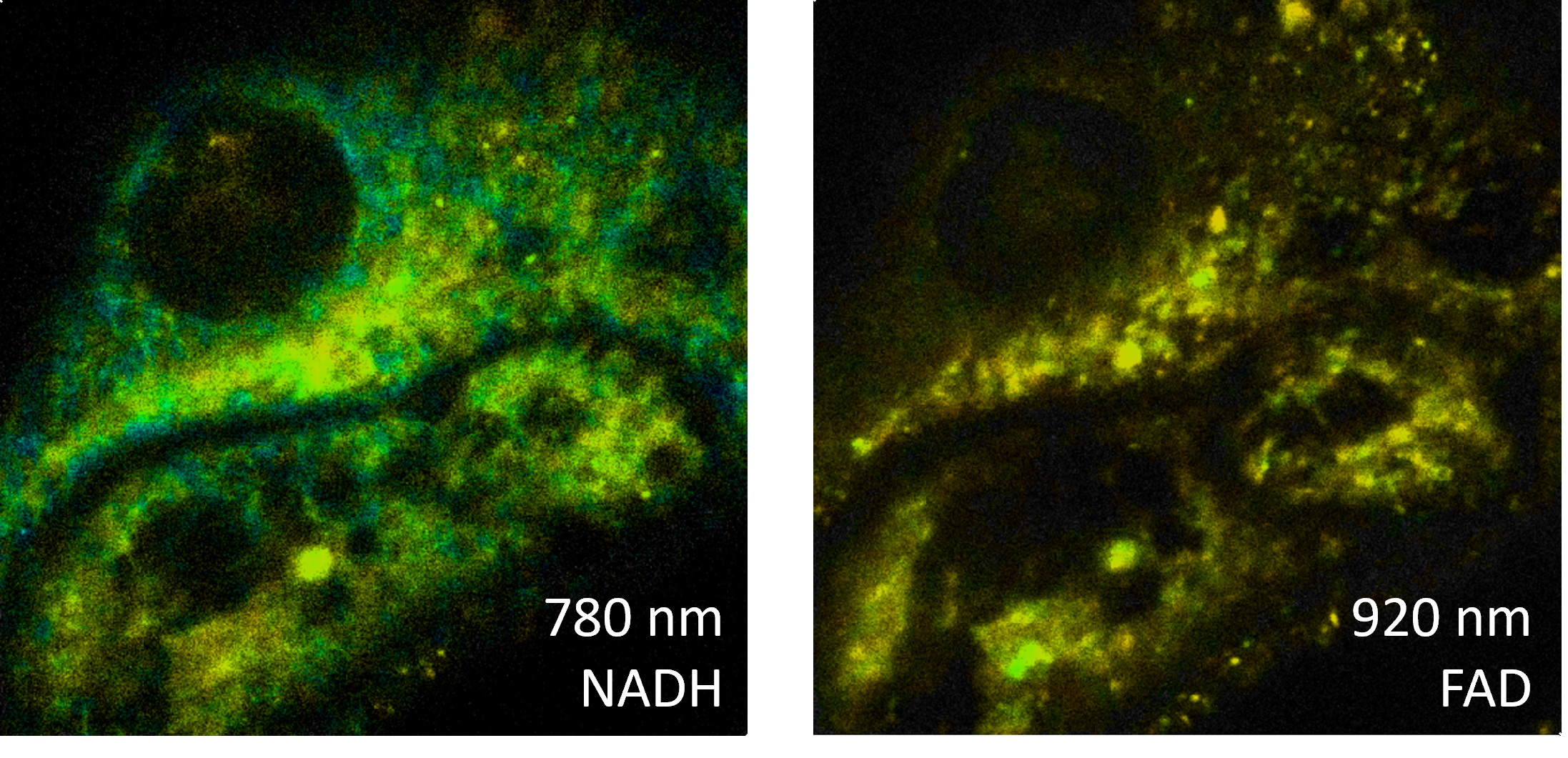
Two-photon fluorescence lifetime imaging (FLIM) can provide insights beyond standard fluorescence microscopy and non-linear microscopy by analyzing fluorescence decay times. In addition, FLIM can be used as a label-free, autofluorescence technique and is therefore non-invasive. For example, simultaneous NADH and FAD decay measurements provide insight into the metabolic state of biological samples.
A fast electronic trigger output from femtosecond fiber lasers serves as a reference for time-correlated single photon counting (TCSPC) electronics in fluorescence lifetime imaging (FLIM) and gated detectors.
→ Typical lasers: Femtosecond fiber lasers at 780, 920 or 1050 nm
Multimodal imaging
Multimodal imaging integrates diverse technological modalities within a unified microscopy platform, thereby facilitating a more comprehensive examination of biological specimens. In a multimodal approach, for instance, signals from molecules such as NADH, FAD, collagen, and elastin are collected simultaneously by two-photon autofluorescence, SHG, and confocal imaging methods. This combination enables researchers to correlate morphological features with functional and metabolic insights in real time.
Multimodal imaging has been demonstrated to maximize information content while minimizing sample preparation and photodamage, thus rendering it a powerful tool for studying complex biological systems, living tissues, and dynamic processes under near-physiological conditions.
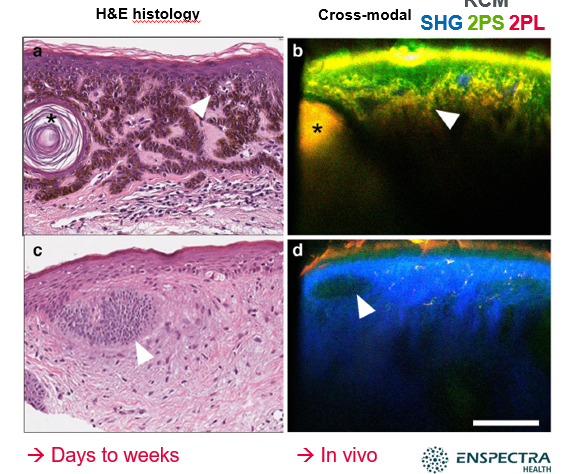
Example: conventional H&E histology versus cross-modal microscopy combining pinhole-free reflectance confocal microscopy (pf-RCM), second harmonic generation (SHG), two-photon autofluorescence, short (2PS) and two-photon autofluorescence, long (2PL). Taken from: Montgomery, et al., Handheld multiphoton and pinhole-free reflectance confocal microscopy enables noninvasive, real-time cross-sectional imaging in skin; Nature (2024)
Why label-free imaging matters
In contrast to traditional fluorescence or immunohistochemical methods, label-free imaging:
-
- Preserves the native biological environment — no dyes, tags, or fixation artifacts.
- Enables long-term, live-cell or in vivo imaging without phototoxicity.
- Provides chemical specificity and structural contrast simultaneously.
- Facilitates real-time diagnosis and quantitative biochemistry in clinical settings.
These advantages position label-free optical imaging as a leading field in biophotonics and clinical applications, thereby facilitating a seamless integration of fundamental research and medical diagnostics. The implementation of these methods includes the use of microscopes in carts, handheld scanners, and endoscopes, thereby facilitating patient access and expediting analysis and research in the following fields:
-
- Life science and biomedical research
- Cancer research and pathology
- Dermatology and skin research
- Regenerative medicine and tissue engineering
- Drug discovery and toxicology
- Clinical and translational research
Simplifying the path to clinical imaging with lasers
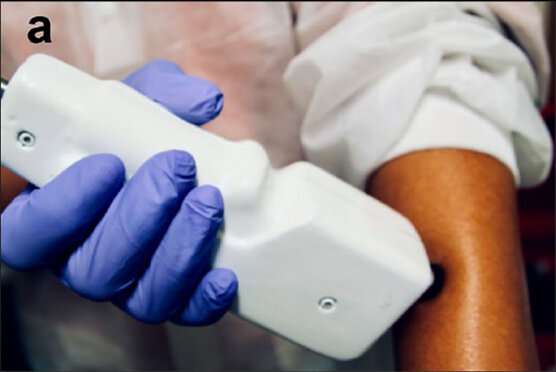
Example of handheld scanner incorporating TOPTICA's FemtoFiber smart 780 PT. Taken from: Montgomery, et al., Handheld multiphoton and pinhole-free reflectance confocal microscopy enables noninvasive, real-time cross-sectional imaging in skin; Nature (2024)
Until recently, the complexity and cost of traditional ultrafast laser systems limited two-photon microscopy to specialized research labs. Fiber-based femtosecond lasers are changing that paradigm — combining turnkey operation and robustness suitable for clinical and translational environments.
This simplification is paving the way for clinical-grade two-photon imaging systems in dermatology, ophthalmology, histology and endoscopy. The new generation of compact, fiber-coupled femtosecond lasers brings the promise of two-photon microscopy from the research bench to the clinical applications.
Scientific Papers
-
Dynamic Retinal Pathology in Glaucoma Progression Revealed by High-Resolution Functional Imaging In Vivo
Yiming Fu, Pham Binh Minh, Sicong He, Yingzhu He, Zhongya Qin, Ting Xie, Jianan Qu; Wiley (2025)
-
Handheld multiphoton and pinhole-free reflectance confocal microscopy enables noninvasive, real-time cross-sectional imaging in skin
Montgomery, et al.; Nature (2024)
-
Metabolic imaging by simultaneous 2-photon FLIM of NAD(P)H and FAD
Becker Wolfgang, Axel Bergmann, Alexander Jelzow, Antje Neubauer, Angelika Rück, Konrad Birkmeier, and Patrick Leisching; Multiphoton Microscopy in the Biomedical Sciences XX, 11244:112440L. International Society for Optics and Photonics (2020)
-
Monocular depth estimation based on deep learning for intraoperative guidance using surface-enhanced Raman scattering imaging
Aniwat Juhong, Bo Li, Yifan Liu, et. al.; Photonics Research (2025)
Application Notes
-
Two-Photon FLIM with a Small Femtosecond Fibre Laser
Becker&Hickl (2023)
-
Two-Photon FLIM with a Femtosecond Fibre Laser
Becker&Hickl (2020)